Draw the structure of Nylon-4,4 (draw 3 repeating units). What monomers would be needed for the formation of Nylon-4,4 (provide the structures)?

Nylon 6,10 is prepared by polymerization of a diamine and a diacid chloride. Draw a structural formula for each reactant and the repeat unit in this polymer.
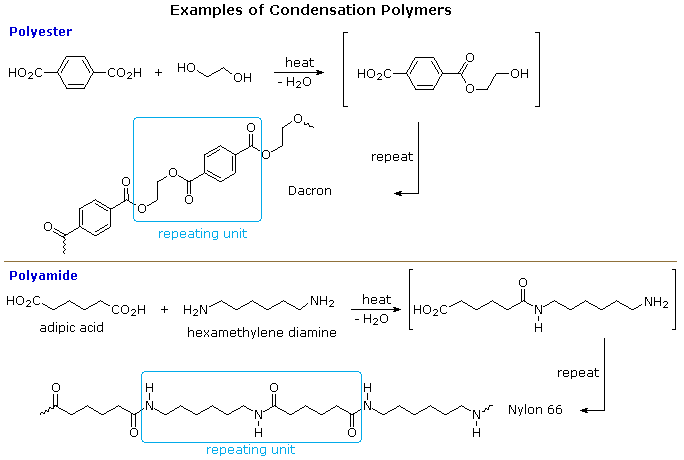
24.6: Step-Growth Polymers—Condensation Polymers - Chemistry LibreTexts
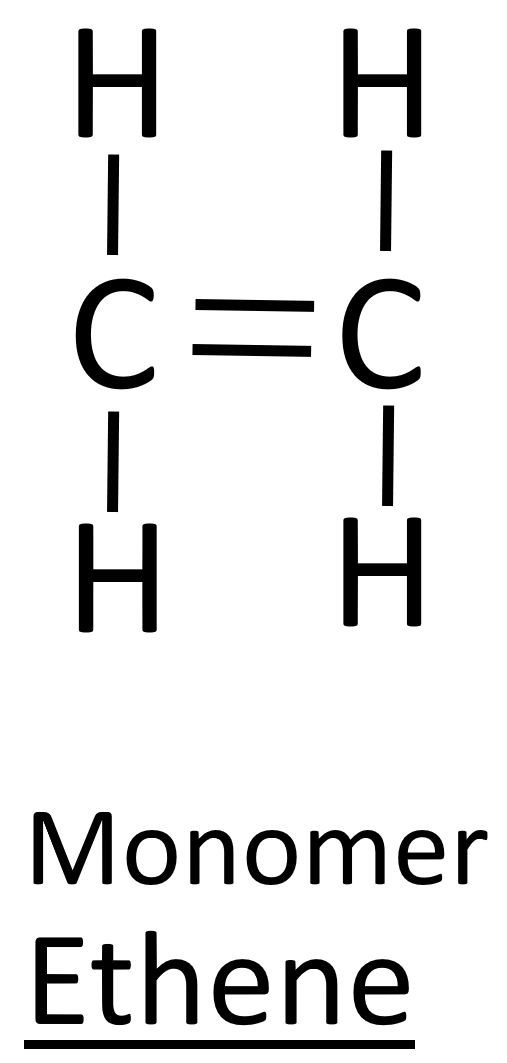
4:45 understand how to draw the repeat unit of an addition polymer, including poly(ethene), poly(propene), poly(chloroethene) and (poly)tetrafluroethene - TutorMyself Chemistry

Draw the structure of Nylon-4,4 (draw 3 repeating units). What monomers would be needed for the formation of Nylon-4,4 (provide the structures)?
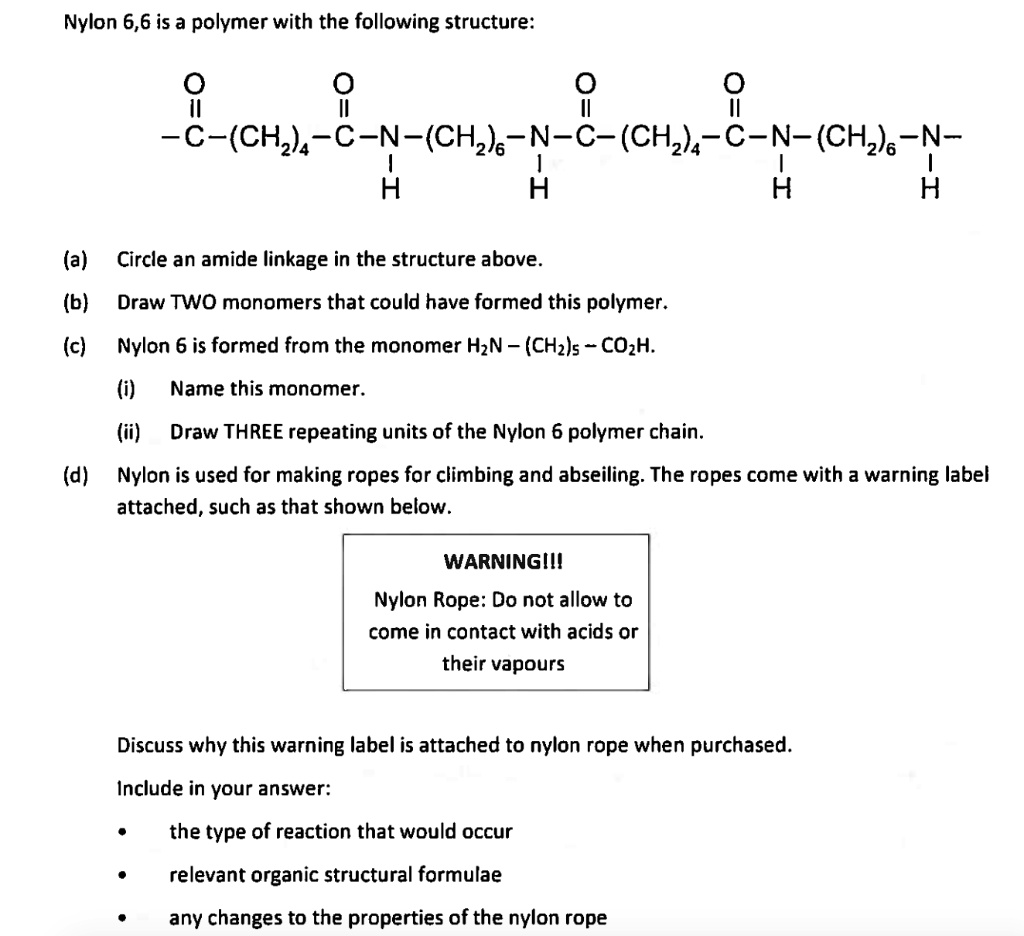
SOLVED: Nylon 6,6 is a polymer with the following structure: c-(CH2)5-C-N-(CH2)-N-c-(CH2)-C-N-(CH2)5-N- H (a) Circle an amide linkage in the structure above. (b) Draw TWO monomers that could have formed this polymer. (c)

INTEXT QUESTIONS 1. Draw the structure of repeating unit in nylon-6,10. 2. What are the monomers used to prepare nylons given below? To O -NH- i) ll +C+CH,),+C - NH 0 ii)
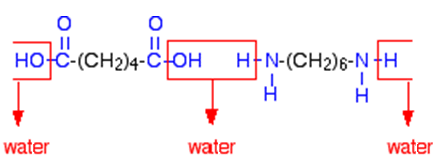
Nylon - MOTM 2010 - VRML version

Man-made fibre - Chemical Composition, Molecular Structure

Percolated Network Structure Formation and Rheological Properties in Nylon 6/Clay Nanocomposites - Mizuno - 2013 - Macromolecular Materials and Engineering - Wiley Online Library
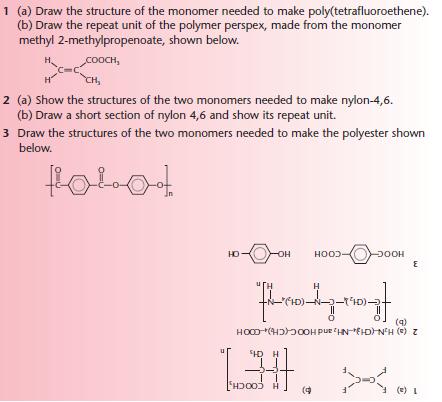
Polymers, a2-level-level-revision, chemistry, aromatics-amines-amino-acids-polymers, polymers

Nylon-6 has the following structure: What is the structure of the monomer unit in this polymer? Show the reaction corresponding to the formation of the polymer.

Solved 2. Write the reaction mechanism (showing arrows) of
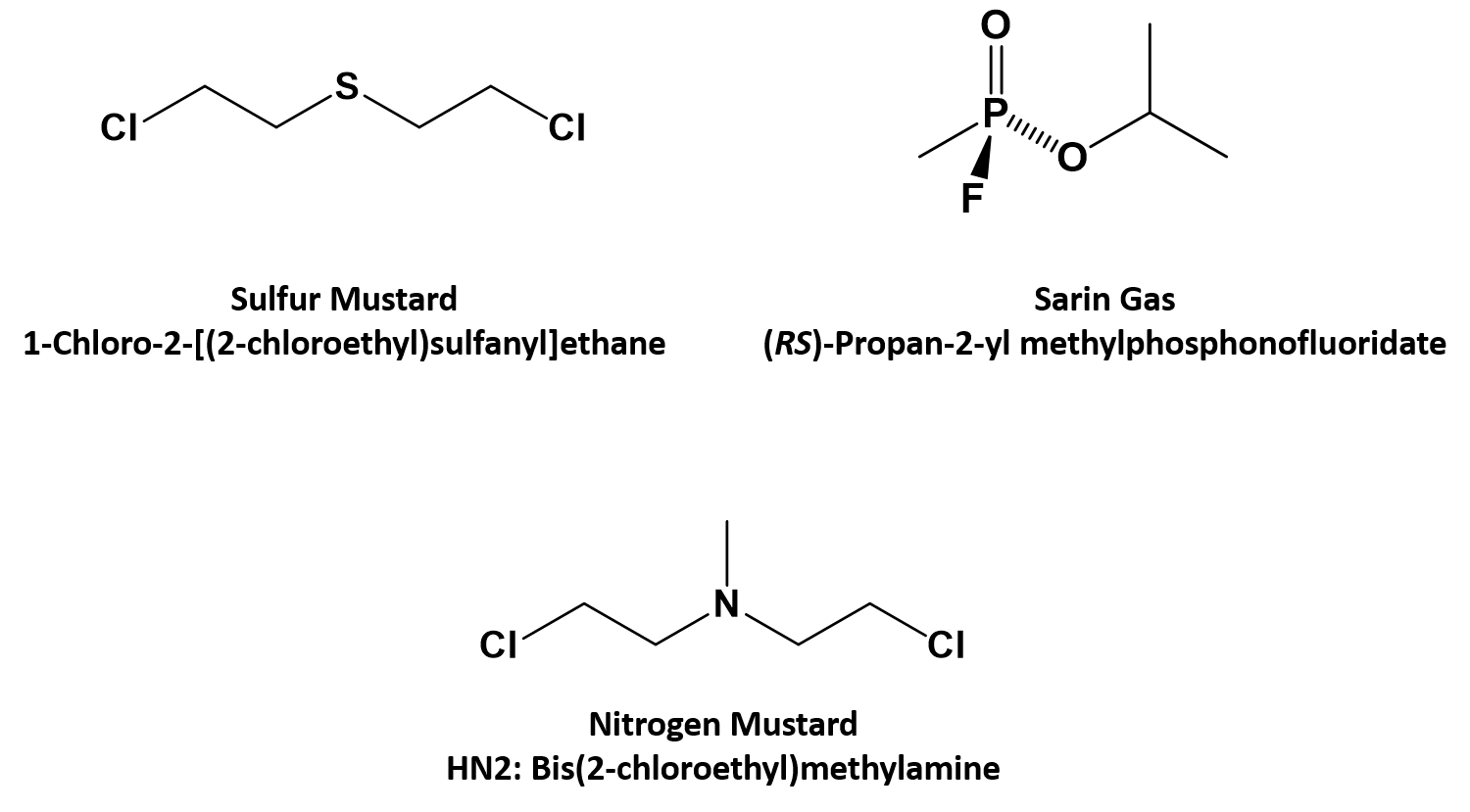
CH105: Chapter 10 - Compounds with Sulfur, Phosphorus, and Nitrogen - Chemistry

Draw the structure of the monomer each of the following polymers:(i) Nylon 6(ii) Polypropene