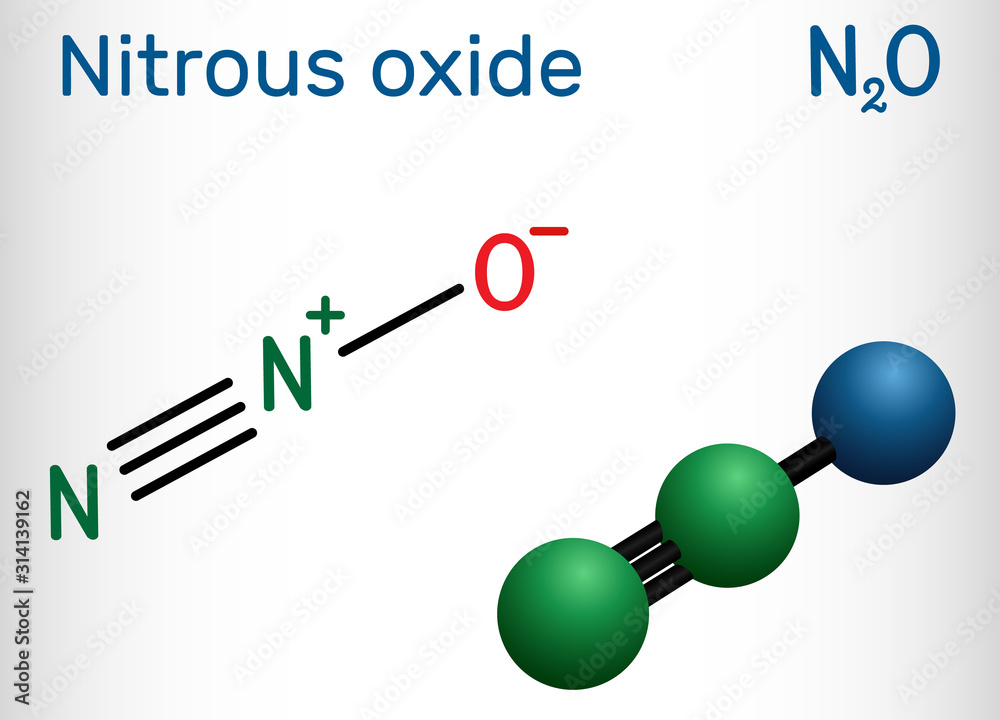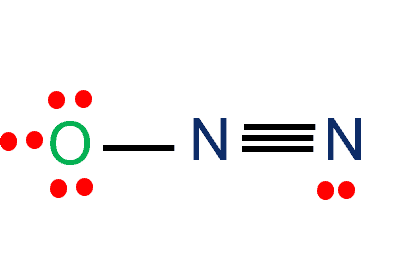
N2O Lewis Structure Nitrous oxide-Laughing Gas - What's Insight
In the N2O lewis structure, there are two atoms of Nitrogen (N) and one oxygen (O) atom. Keep reading to know more about N2O and laughing gas.

Medicina, Free Full-Text

Frustrated Lewis Pair Stabilized Phosphoryl Nitride (NPO), a Monophosphorus Analogue of Nitrous Oxide (N2O)
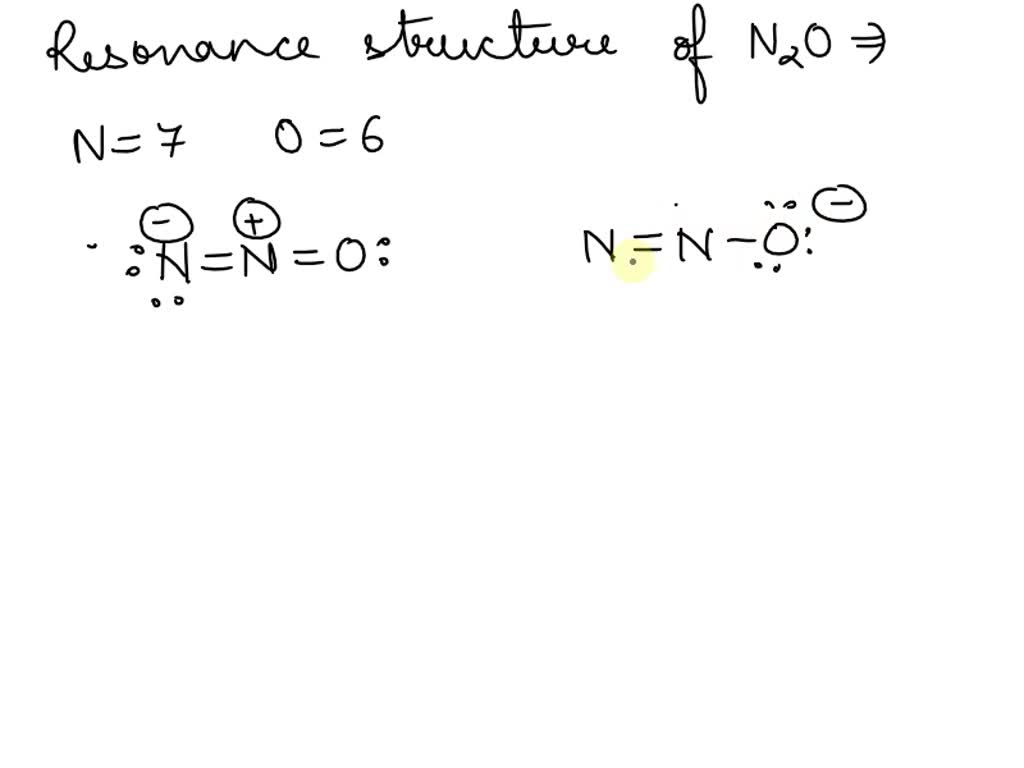
SOLVED: Called laughing gas, nitrous oxide (N2O) is sometimes used by dentists as an anesthetic. Given the connections N-N-O draw two electron-dot resonance structures for N2O.

Increasing recreational nitrous oxide use: Should we worry? A narrative review - Jan GC van Amsterdam, Ton Nabben, Wim van den Brink, 2022

Nitrous oxide emissions from soils: how well do we understand the processes and their controls?

Nitrous oxide - Wikipedia

Nitrous oxide-induced slow and delta oscillations - ScienceDirect

Two-Pore-Domain K+ Channels Are a Novel Target for the Anesthetic Gases Xenon, Nitrous Oxide, and Cyclopropane

N2O Lewis Structure Nitrous oxide-Laughing Gas - What's Insight

Three non-equivalent resonance structures are possible for nitrous oxide ( N2O), laughing gas. a. Draw the three resonance structures and indicate the formal charge on each atom in each resonance structure. b. Based

Chemistry Net: Lewis Dot Structures - Simple Procedure for writing Lewis Structures of Nitrous Oxide (N2O)